Schedule an appointment to get vaccinations for flu TDAP shingles and other conditions. Vaccine appointments for this site are currently released on Thursday mornings at 9 am and go quickly.
 Covid 19 Vaccine Clinics District 4 Public Health Georgia Public Health Department
Covid 19 Vaccine Clinics District 4 Public Health Georgia Public Health Department
Tells you if you are currently eligible to receive a vaccine.

Vaccine appointments near me. For vaccine appointments for individuals ages 16- or 17-years old use the Vaccine Type drop down menu to search for vaccination locations with Pfizer. Vaccines for you and your family We have the CDC-recommended vaccines you need administered by an immunization-trained pharmacist. This tool does not allow you to schedule a vaccination appointment.
If youre an eligible unpaid carer but you are not able to book an appointment speak to your GP surgery. COVID-19 Vaccine Sites in Maine Maine residents age 16 and older are currently eligible for COVID-19 vaccination. Pfizer Saturday May 1.
Find vaccines near you. Minors will need a parentguardian consent to register for a vaccine appointment. Schools religious centers travel clinics and workplaces can also provide these vaccines under some circumstances.
Have 2 doses of the COVID-19 vaccine at 2 appointments book both appointments at the same time get the 2nd dose 11 to 12 weeks. Below youll find information and tools to help you find out where to get vaccinated near youand some tips for making an appointment. Use the tool at that time for the best chance of getting an appointment.
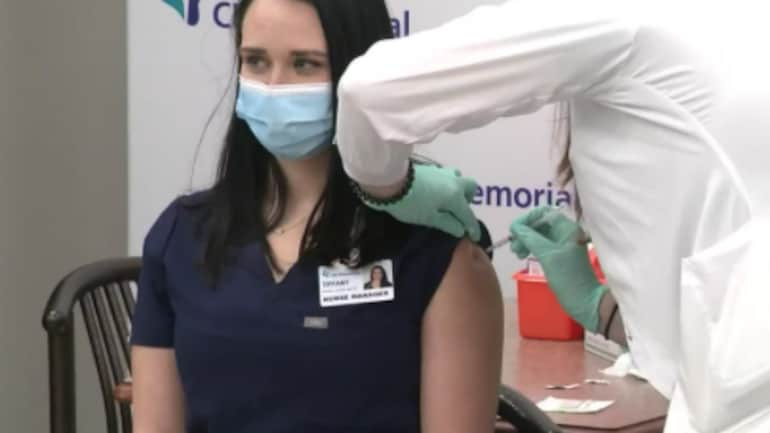
If you are eligible you may hear directly from your health care provider about getting a vaccine or you may contact the locations below for an appointmentAll sites require an appointment. Please keep in mind there is still a high demand for appointments and a limited number become available at once. Depending on which vaccine you receive you will be prompted to schedule a second dose if needed.
Providers have to be registered to receive the vaccine so make sure your chosen provider is. Please keep in mind there is still a high demand for appointments and a limited number become available at once. Use the COVID-19 vaccine eligibility checker to find out when you can receive a COVID-19 vaccine book an appointment if you are eligible or register your interest.
AND Have your parent or legal guardian schedule your appointment to provide consent to receive the vaccine. If you need help scheduling an appointment call the Maine COVID-19 Community Vaccination. Schedule an Appointment to receive the Covid vaccine at a store near you.
Includes information on eligibility vaccine types available registration systems and unique registration links that have been made available. Given limited supply of all three types of COVID-19 vaccines at this time we recommend that you receive whichever. Select an appointment from a site offering the Pfizer-BioNTech vaccine only.
COVID-19 vaccines Is it true. Find out more about who is eligible to have a COVID-19 vaccination. Flu vaccines and other routine vaccines will be back on the site in the near future.
Helps you create an account and find a vaccination provider when you are eligible to receive a vaccine. Find out if you are eligible to receive the COVID-19 vaccine. Please do not contact your local CVS pharmacy directly with questions about the COVID-19 vaccine.
Walgreens is now booking COVID-19 vaccination appointments online. Flu or other routine vaccines are usually available at private doctors offices health department clinics urgent care clinics and pharmacies. Vaccine providers can include hospitals urgent care centers doctors offices and pharmacies.
Additional information provided to assist in preparing for your appointment and next steps after vaccination. Get vaccinated at a store near you. CVS COVID-19 Resource Center Español.
People Age 16 or 17 can only receive the Pfizer vaccine. Walgreens recommends that you check the Walgreens pharmacy website for updates and dates when more vaccines will become available. Schedule a vaccination Get COVID-19 vaccine info.
Go here for vaccine information and to find a vaccine near you. CVS recommends that you check the CVS pharmacy website for updates and dates when more vaccines will become available. If you visit a no-appointment vaccination site a parent or legal guardian must be with you to provide consent in order for you to receive the vaccine.
Visit wwwyourspotyourshotncgov to learn more about COVID-19 vaccines. Signs you up to receive important vaccine eligibility updates. Listing of appointments for COVID-19 Vaccination in the Los Angeles County.
Pfizer ages 16 Sunday April 25. Find accurate evidence-based answers to questions about COVID-19 vaccines. If you have questions about this tool or need help understanding the appointment-making process call the NC COVID-19 Vaccine Help Center at 888-675-4567.
You can get most recommended vaccines at your doctors office and many recommended vaccines are also available at local pharmacies health centers health departments and travel clinics.











/GettyImages-1251002737-866d85dbf6684362842555918ff1eae2.jpg)